There are growing concerns worldwide that Russian invaders may cause a catastrophe at the Zaporizhia Nuclear Power Plant, which is under their control, resulting in significant radiation contamination of Ukrainian territory… And we know that there is a considerable ionizing radiation in space that poses a threat to astronauts. However, there is a significant difference between what to fear in each of these cases.

Is all radiation lethal?
After the Russian savages detonated the dam of the Kakhovka Hydroelectric Power Station and caused an environmental disaster, many people became fearful that the Zaporizhia Nuclear Power Plant would be their next target. Since it is under their control, the invaders could attempt a serious accident at any time.
As a result, more and more people are starting to think about the dangers of radiation. However, the majority of the population still does not understand what exactly they should fear. Therefore, it is worth examining this question in more detail.
The term “radiation” itself, as it is commonly used, is completely incorrect. This Latin word simply means “dispersion of rays ” and is used in physics to denote any particles that emanate from a particular source. In this sense, sunlight from the Sun is also radiation, although it is not usually considered dangerous.
What everyone is rightfully afraid of is properly called ionizing radiation. It includes all particles that have energy greater than 10 electron volts. Upon collision with matter, these particles are capable of breaking chemical bonds, transforming molecules into ions, hence the name.
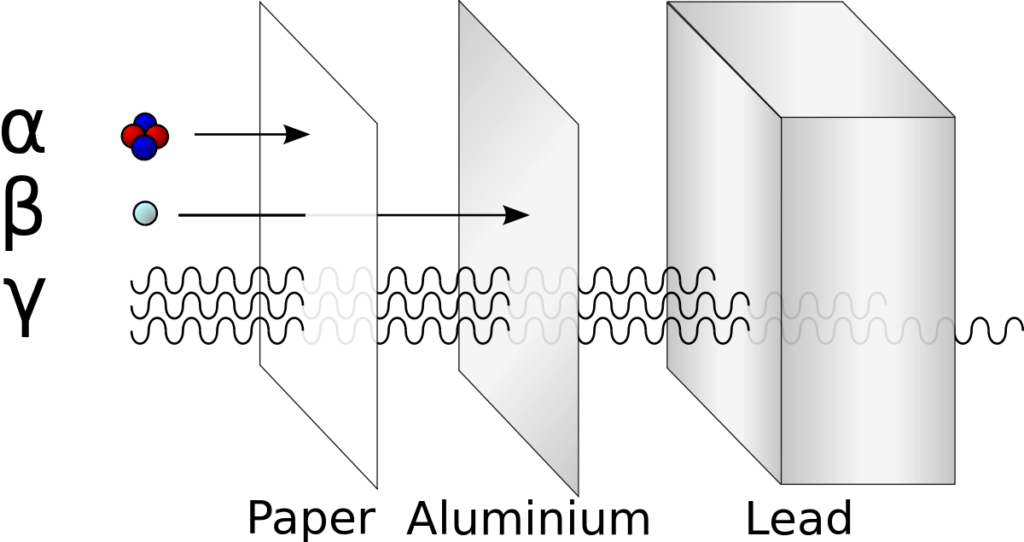
Ionizing radiation includes streams of protons, neutrons, muons, electrons, and even certain ions, such as alpha particles (helium nuclei). It also includes the high-energy portion of the electromagnetic spectrum: “hard” ultraviolet, X-rays, and gamma radiation. These types of radiation can indeed cause significant harm.
Radiation Dose
However, when it comes to streams of charged particles, it is not as straightforward. The energy carried by each individual particle cannot lead to any fatal consequences in a large system such as the human body. What matters here is the amount of energy absorbed from ionizing radiation by a volume of material over a certain period of time. This quantity is generally referred to as radiation dose.
But even that is not decisive. The amount and energy spectrum of ionizing radiation particles vary chaotically. Moreover, they do not transfer all their energy to the substance they encounter. Therefore, there are several different kinds of doses.
The first one is called the exposure dose. It measures how much ionizing radiation ionizes molecules of dry air. Since ions are charged particles, the process itself is essentially the accumulation of charge. Thus, the exposure dose is determined as the total charge accumulated by ions in one kilogram of air as a result of irradiation and is measured in coulombs per kilogram. However, a much more common unit of measurement is the roentgen, which is equal to 2.58 × 10⁻⁴ coulombs per kilogram.

People most commonly encounter this dose. Every time we hear in a weather forecast that the radiation background is a certain amount of micro-roentgens per hour, we learn about the exposure dose that the air in a specific region receives in a given unit of time.
However, solid objects, including human bodies, absorb ionizing radiation differently than gases. When particles penetrate them, the initial impulse leads to a series of processes that are quite difficult to account for. Therefore, a different type dose, called the absorbed dose, is used for them. It is defined as the amount of energy that radiation has transferred to a unit of mass. The main unit of measurement for this dose is the gray, which is equal to one joule of energy per kilogram of mass. The rad, which is one hundredth of a gray, is also used.
What is a safe dose?
When it comes to the impact of radiation on living beings, a third type of dose is used called the equivalent dose. Its existence is due to the fact that different types of ionizing radiation have varying effects on the materials they interact with, even if they have the same energy. The heavier the particle, the more damage it causes.
Therefore, the equivalent dose is determined by multiplying the absorbed dose by a certain coefficient. For photons of ultraviolet, X-ray, and γ-radiation, as well as electrons and muons, the coefficient is equal to one. This means that for these types of radiation, the equivalent dose is equal to the absorbed dose.
For heavier protons, the coefficient is 2, and for α-particles, it is 20. For neutrons, the coefficient is variable and ranges from 2.5 to 20.7. Numerically, the equivalent dose is also measured in joules per kilogram, but in this case, the unit is called the sievert (Sv). There is also a non-system unit called the rem (roentgen equivalent man), which is equal to one hundredth of a sievert (0.01 Sv).
The term sievert (denoted as Sv) is usually used when discussing the health damage that a person may receive from visiting an X-ray room, space travel, or accidents at nuclear power plants.
It is believed that on average, each person on Earth receives around 3 millisieverts (mSv) of radiation per year, although some individuals may receive significantly more. This radiation accumulates over a lifetime. It is possible that some health conditions that arise in old age are related to the accumulation of this natural radiation, but we cannot escape it.
However, it is quite possible to accelerate the onset of these diseases by being in close proximity to sources of ionizing radiation. Therefore, people whose activities are associated with such sources carefully monitor the accumulated dose.
In extreme cases, the intensity of ionizing radiation becomes so high that it can cause immediate harm to the organism. This is referred to as the fourth kind of dose of radiation — the effective dose.

As with the previous two, it is measured in sieverts but is determined separately for each organ of the human body. This is because strong radiation generates a huge number of ions that trigger completely abnormal chains of biochemical reactions, causing the organ to start self-destructing in a unique way.
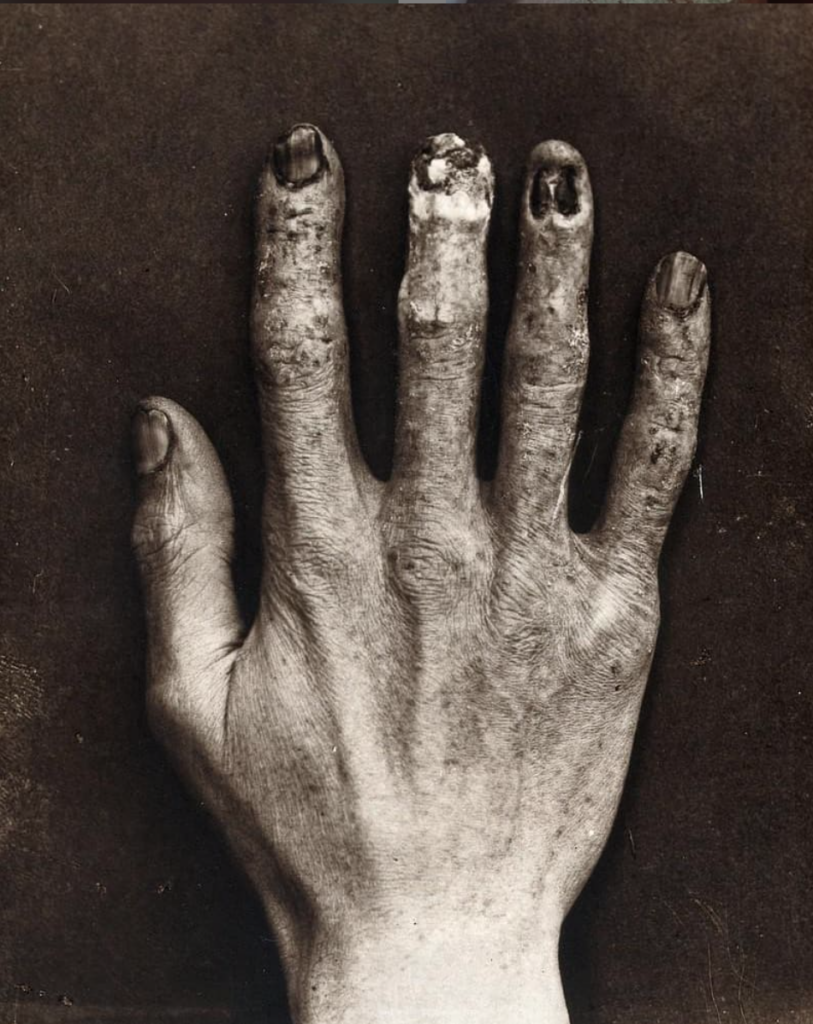
In extreme cases, phenomena of induced radioactivity are observed. Due to the action of ionizing radiation, atoms that absorbed its energy themselves become a source of dangerous radiation. In such cases, it almost always ends with acute radiation sickness and a rapid and painful death.
That is why determining a safe radiation dose for a specific individual is an extremely challenging task. Scientists have a relatively good understanding of the cases where severe fatal illnesses are practically unavoidable. However, research is still being conducted regarding the effects of low doses. It is believed that the accumulation of 1 Sv leads to radiation sickness, 4 Sv can quickly and highly likely kill a person, and at 8 Sv, death is guaranteed.
Radiation in space
Ionizing radiation is one of the main obstacles in space exploration. It is everywhere in space. The Earth’s atmosphere absorbs the majority of it, so out of our annual exposure of 3 mSv, we only receive 0.39 mSv from cosmic rays. However, for commercial airline pilots, this value can reach 2.2 mSv.
Astronauts on the International Space Station (ISS) receive a dose that is 200 times higher than people on Earth—0.5-0.7 Sv per day. After spending several years in orbit, the same 1 Sv is accumulated in their bodies, after which doctors begin to observe signs of radiation sickness.

All of this occurs in relatively safe areas of outer space in terms of ionizing radiation. There are much more dangerous places, such as the radiation belts of giant planets, the vicinity of neutron stars, and black holes. The intensity of radiation there is so high that a lethal dose can be received in a matter of seconds.
One peculiarity of radiation in space is that there are always many high-energy particles in a vacuum, and they come in all types—from photons to helium nuclei and even heavier elements. The sources of their radiation are mostly located very far away, although they are incredibly numerous themselves. The closest of those source is the Sun.
For astronauts in low Earth orbit, our star poses the greatest danger. Solar flares, which often “launch” not only electromagnetic waves but also α-particles towards our planet, have a coefficient of 20 according to the equivalent dose calculation. Fortunately, most of these particles are well absorbed by the hulls of spacecraft and orbital stations.
However, it is very difficult to shield against high-energy electromagnetic radiation. How this problem will be solved in the future is still unclear. Perhaps efficient means of shielding will be invented, or drugs that prevent the development of radiation sickness will help, or human DNA itself will be altered in a way that allows us to live in space.
What will happen during a nuclear power plant accident?
At first glance, during a nuclear power plant accident — whether caused by a mistake or malicious intentions — we will all find ourselves in the role of astronauts. There will be a lot of radiation around, and we will all have to deal with it. However, there are significant differences in reality.

Unlike in space, on Earth, there is the surrounding environment — soil, water, and air. It absorbs radiation to some extent. Therefore, the dispersion of particles will not be infinite. Ionizing radiation will get relatively localized around its sources.
The problem lies in the sources themselves. They include everything contained inside the reactor: nuclear fuel, its decay products, even pipes and structural materials have a certain induced radioactivity. We have seen what happens when all this spreads over a large area in the case of the Chernobyl disaster. In places where fragmented sources of radioactive isotopes entered the soil, radiation level meters start going crazy many years after the accident.
Staying on the ground in such areas is extremely dangerous for health because it is where a huge number of high-energy particles are born. How many? This is indicated by a physical quantity called radioactivity. Its unit of measurement is the becquerel, which represents one decay event per second. Usually, the birth of one radioactive particle occurs during this process.

So, one gram of uranium-235, which is used as fuel in nuclear power plants, has a radioactivity of 80 kilobecquerels. This means that its tiny speck generates 80,000 radioactive particles per second. And a significant portion of them will reach a person standing or sitting nearby.
However, there can be an even worse scenario when the radioactive material enters the body through the respiratory tract or stomach. In this case, it can remain in certain organs for years, and then all the high-energy particles reach that person. Becquerels directly convert into sieverts.
Disasters may not occur
In case of a nuclear power plant accident, experts are primarily concerned about the spread of radioactive materials. However, it is not as easy to achieve this in the case of the Zaporizhzhia Nuclear Power Plant as it was with Chernobyl. The WWER-1000 reactors operating there simply cannot explode like it happened in 1986. The destruction was caused by the rupture of pipes containing overheated gas, which destroyed the reactor in the situation where the reaction could not be stopped.

In the case of WWER-1000, such a situation simply cannot occur. Even if, for some reason, the cooling system fails (which is not so easy to attain even deliberately), the reactor will not explode but melt. And even in this case, the threat of radioactive substances escaping beyond its boundaries will be minimal. The thing is, this type of reactor, unlike the RBMK reactors installed at the Chernobyl Nuclear Power Plant, has a strong containment structure specifically designed to retain hazardous materials in the event of an accident.
Of course, if the invading species firmly decide to create a global catastrophe, no one can provide 100% reliable protection against their “ingenuity.” However, if they attempt to radically change the technological processes at the plant or place tons of explosives under the reactors, it will be extremely difficult to conceal it. One can only hope that in such a scenario, the international community will find a way to neutralize the threat before it becomes a reality.
